Chemistry OF MiNt

Uncovering the mystery of mint as well as any other matter should start with a chemical study to find out its composition.
This chemical minty page will inform you about the composition of mint and the chemistry of its main component : MENTHOL
I- Chemical Composition of MINT:
The chemical components of peppermint oil are menthol, menthone, cineole, methyl acetate, methofuran, isomenthone, limonene, b-pinene, a-pinene, germacrene-d, trans-sabinene hydrate and pulegone. Menthol in mint is responsible for the cooling effect of mint.

II- Chemistry of MENTHOL:
A- Molecular formula of Menthol: C 10 H 20 O
B- Structural formula of Menthol:
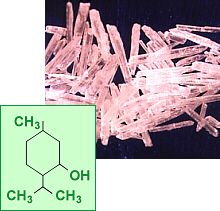

C- Functional groups of Menthol: In the natural compound, the isopropyl group is in the trans orientation to both the methyl and hydroxyl groups.
D- Reactions: Menthol reacts in many ways like a normal secondary alcohol. It is oxidised to menthone by oxidising agents such as chromic acidor dichromate,though under some conditions the oxidation can go further and break open the ring. Menthol is easily dehydrated to give mainly 3-menthene, by the action of 2% sulfuric acid. PCl5 gives menthyl chloride.

III- Physical properties of MENTHOL:
|
Molecular formula
|
C10H20O
|
|
Molar mass
|
156.27 g/mol
|
|
Appearance
|
White or colorless
crystalline solid
|
|
Density and phase
|
0.890 g/cm3, solid
(racemic or (−)-isomer)
|
|
Solubility in water
|
Slightly soluble, (−)-isomer
|
|
In ethanol, diethyl ether, acetone, chloroformacetic acid, hexane
|
Soluble
|
|
Melting point
|
36-38 °C (311 K), racemic
35-33-31 °C, (−)-isomer
|
|
Boiling point
|
212 °C (485 K)
|
|
Odor
|
Peppermint oil
|
|
Density
|
0.89 g/l
|
|
Vapor Pressure (mm Hg)
|
0.8 at 21C (70F)
|
|
Chiral rotation *D
|
-50° at 18 °C, 10% Ethanol Solution
|
|
Main Hazards
|
Irritant, flammable
|
|
Thermodynamic Data
|
Phase behavior : Solid, liquid, gas
|
|
Related Alcohols
|
Cyclohexanol, Pulegol,
Dihydrocarveol, Piperitol
|
|
Related Compounds
|
Menthone, Menthene, Thymol, p-Cymene,
Citronellal
|
Note: The data are given for materials in their standard state (at 25°C, 100 kPa).

IV- Stereochemistry of MENTHOL:
The Menthol molecule contains 10 carbon atoms, 3 of these carbon atoms are chiral (that is bonded to 4 different groups of atoms). This results in 8 stereoisomers for Menthol which are: (L)-menthol, (D)-menthol , (L)-isomenthol, (D)-isomenthol, (L)-Neomenthol, (D)-Neomenthol, (L)-Isoneomenthol and (D)- Isoneomenthol.
Stereoisomers are isomers which have their atoms connected in the same sequence but differ in the way the atoms are oriented in space - i.e. the difference between two stereoisomers lies only in the three dimensional arrangement of atoms.
The follow figure shows the condenesed structural formulas of the 8 stereoisomers of menthol. The straight line bonds are lining in the plane of this page. The dark bonds are lining toward you and the dash bonds are lining away backward.

.JPG)
If you noticed the letters (L) and (D) before the name of the stereoisomers in the text but in the figure the names are preceeded by (-) or (+). Don't get confused, it's simple.
(L) means levorotatory (D) means dextrorotatory. The (L) stereoisomer rotates the light to the left and it can be symbolized by (-) however the (D) stereoisomer rotates the light to the right and it can be symbolized by (+). (+) and (-)menthol are mirror images and not superimposable , thus they are called enantiomers .
Stereoisomers of any substance as well as menthol have different properties.
However, the enantiomeric isomers (d and l)have identical physical properties (apart from their specific rotation), but the racemates ( racemic mixture: dl) differ from the optically active forms in, for example, their melting points.
Note: A racemic mixture composed of D-isomer and its mirror image L-isomer each of 50%. That is, it is a mixture of enantiomers. This mixture is not able to rotate light, thus it is optically inactive.
.jpg)
The table below shows the melting point, boiling point and angle of rotation of polarized light in 20% ethyl alcohol solution of one optically active and the racemic form each stereoisomer of menthol.
|
Stereoisomer
|
Melting point
oC
|
Boiling point at 760 Mm, oC
|
(α) in 20% ethyl alcohol
|
|
l- Menthol
|
42.0
|
212
|
-50.0
|
|
d- Menthol
|
43.0
|
216.5
|
+ 50.0
|
|
dl- Menthol
|
38.0
|
216.5
|
0
|
|
d-Neo-Menthol
|
-15.0
|
211.7
|
+20.0
|
|
dl-Neo-Menthol
|
52.0
|
211.7
|
0
|
|
d-Iso-Menthol
|
82.5
|
218.6
|
+26.0
|
|
dl-Iso-Menthol
|
63.5
|
218.6
|
0
|
|
d-Neo-Iso-Menthol
|
-8.0
|
214.6
|
+2.0
|
|
dl-Neo-Iso-Menthol
|
13.5
|
214.6
|
0
|
So Which Stereosomer Of Menthol Is Responsible For The Fresh Cooling Effect Of Mint?

Before answering this question, let’s have an overview for some physical properties of the 8 stereoisomers of menthol in the following table

|
Characteristics of Each Stereoisomer of Menthol
|
|
(-) neomenthol
|
(+) neomenthol
|
|
Minty - musty – fresh – earthy campherous – some cooling
Taste threshold = 600 ppb
Cooling threshold = 25000 ppb
|
Minty - musty – fresh – some cooling - sweet
Taste threshold = 500 ppb
Cooling threshold = 2500 ppb
|
|
(-) menthol
|
(+) menthol
|
|
Very cooling – sweet – fresh - minty
Taste threshold = 400 ppb
Cooling threshold = 800 ppb
|
Fresh – cooling – minty with musty – bitter
Taste threshold = 300 ppb
Cooling threshold = 3000 ppb
|
|
(-) isomenthol
|
(-) isomenthol
|
|
Musty - sweet– hay – earthy camphoraceous – herbaceous – slight cooling.
Taste threshold = 600 ppb
Cooling threshold = 30000 ppb
|
Musty – woody – fresh – carrot minty - earthy camphoraceous –slight cooling.
Taste threshold = 700 ppb
Cooling threshold = 7000 ppb
|
|
(-) neoisomenthol
|
(+) neoisomenthol
|
|
Minty – earthy – woody – camphoraceous – sweet – minty – slight cooling
Taste threshold = 1000 ppb
Cooling threshold = 6000 ppb
|
Minty – earthy – woody – camphoraceous – carrot – minty – herbaceous - very little cooling
Taste threshold = 200 ppb
Cooling threshold = 25000 ppb
|
Note: ppb means part per billion
If you compared the cooling threshold of the stereoisomers of menthol you can find the answer.
Yes  it is (-) or (L) menthol who has the lowesr cooling threshold and is responsible for the cooling effect of pepper mint.
it is (-) or (L) menthol who has the lowesr cooling threshold and is responsible for the cooling effect of pepper mint.
To Know more About Stereochemisty and chirality You Can view this work sheet by just clicking on this link

 it is (-) or (L) menthol who has the lowesr cooling threshold and is responsible for the cooling effect of pepper mint.
it is (-) or (L) menthol who has the lowesr cooling threshold and is responsible for the cooling effect of pepper mint. Chirality of molecules
Chirality of molecules 








Comments (0)
You don't have permission to comment on this page.